
Water is among the most commonly utilized raw materials used in the manufacture of pharmaceuticals, making its quality directly tied to product safety and regulatory compliance. An improperly designed or poorly monitored water system may lead to microbial contamination, failed audits, product recalls, production downtime costs and costly production downtime – thus justifying why purified water system validation plays such an integral part of pharmaceutical operations worldwide. As global regulatory agencies increase GMP compliance requirements for pharmaceutical manufacturers, validation, testing and ongoing monitoring must ensure their purified water systems consistently meet strict quality and performance standards for pharmaceutical operations worldwide.
Purified Water System in Pharmaceutical Manufacturing
Purified water (PW) system is a high-quality water treatment system that is designed to remove microorganisms, contaminants as well as dissolved solids and organic impurities that are present in source water.
Water purified is often utilized in:
- Production of pharmaceuticals
- Cleaning equipment
- Ingredient preparation
- Lab applications
- Cleaning-in-place (CIP) systems

Main Components of a Purified Water System
| System Component | Main Function |
| Pretreatment System | Eliminates hardness, chlorine and suspended particles |
| Reverse Osmosis (RO) | Eliminates dissolved salts and other contaminants |
| EDI System | Further cleanses Ionic contaminants |
| UV Sterilization | Limits the growth of microbial species |
| Storage Tank | Stores safe water purified |
| Distribution Loop | Water is delivered throughout the facility |
Modern pharmaceutical facilities usually utilize advanced technology, like those of the Molewater Water Purification System to ensure the production of stable pharmaceutical water and long-term reliability of operation.
Purified Water System Validation for GMP Compliance
In the manufacturing of pharmaceuticals water is considered an “critical utility” because it directly affects product quality.
The regulatory agencies require manufacturers to prove the following
- Be consistent in operation
- Maintain control of microbiological activity
- Prevent the risk of contamination
- Produce water that meets pharmacopeia standards
Without a proper purified water system validation, businesses risk:
- FDA warning letters
- GMP audit failures
- Product contamination
- Production shutdowns
Water System Risks in Pharmaceutical Facilities
In systems that aren’t maintained, problems can arise:
- Biofilm creation
- Microbial contamination
- Legs that are dead in pipes systems
- Organic contamination buildup
These issues could compromise the safety of products as well as health for patients.
Regulatory Expectations
Important pharmaceutical regulations that relate to water systems are:
- FDA CGMP specifications
- EU GMP Annex 1
- WHO GMP guidelines
- USP <1231> Water for Pharmaceutical Purposes
These standards emphasize:
- Hygienic system design
- Continuous monitoring
- Validation documentation
- Microbial control routine
Key GMP Requirements for Purified Water Systems
To achieve GMP compliance involves more than just setting up a purification system. The entire system needs to be designed, monitored and maintained in accordance with the pharmaceutical standards.
Hygienic System Design
A well-designed system can help to reduce the risk of microbial growth.
Important design requirements include:
- SS316L stainless steel pipes
- Sanitary welding
- Minimal dead legs
Proper slope of the pipe and drainage
Dead legs — or sections of piping that are stagnant are the most common source of microbiological contamination.
Continuous Circulation
The majority of pharmaceutical water loops run constantly to ensure water flow and limit the growth of bacteria.
Maintaining a proper loop speed helps:
- Prevent stagnation
- Reduce biofilm formation
- Improve sanitization effectiveness
Water Quality Monitoring
Key parameters typically monitored are:
| Parameter | Purpose |
| Conductivity | Tests for ionic contamination |
| TOC (Total Organic Carbon) | It detects organic impurities |
| Microbial Count | Controls the levels of bacteria |
| Temperature | Helps to control microbial growth |
Monitoring systems that monitor real-time are increasingly utilized to improve the operational reliability.

Documentation and Traceability
Regulators require complete documentation for:
- SOPs
- Maintenance records
- Calibration logs
- Sanitization procedures
- Validation reports
Accurate documentation is essential during FDA and GMP inspections.
Three Main Stages of Purified Water System Validation
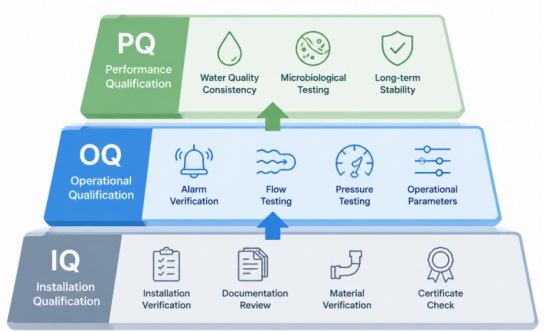
Installation Qualification (IQ)
Installation Qualification confirms that the water system is installed according to approved engineering and GMP specifications.
IQ activities usually include:
- Verifying piping materials
- Reviewing equipment documentation
- Checking P&ID drawings
- Confirming instrument calibration
- Inspecting sanitary welding quality
Typical IQ Checklist
| IQ Verification Item | Purpose |
| Material Certificates | Verify GMP-compliant materials |
| Piping Inspection | Confirm hygienic installation |
| Calibration Records | Ensure instrument accuracy |
| Equipment Verification | Match approved specifications |
IQ establishes the foundation for future operational testing.
Operational Qualification (OQ)
Operational Qualification verifies that the system performs properly under defined operating conditions.
OQ testing commonly includes:
- Alarm verification
- Flow testing
- Pressure testing
- Sanitization procedure testing
- Conductivity monitoring
Engineers often test:
- Startup conditions
- Shutdown recovery
- Worst-case operating scenarios
The goal is to confirm stable operational performance.
Performance Qualification (PQ)
Performance Qualification demonstrates that the system consistently produces purified water meeting pharmaceutical quality standards during routine operation.
PQ is often the most important stage of purified water system validation.
It usually includes:
- Long-term sampling
- Microbial testing
- Conductivity analysis
- TOC monitoring
- Trend analysis
Many pharmaceutical facilities follow a three-phase PQ approach:
- Intensive daily monitoring
- Reduced-frequency testing
- Long-term routine monitoring
This helps confirm long-term system stability.
Common Purified Water System Validation Failures
Many GMP audit findings are attributed to inadequate water system management.
Dead Legs in Distribution Loops
Dead legs allow stagnant waters to collect, creating ideal conditions for the growth of microbial species.
Poor Sanitization Procedures
Inadequate chemical or thermal cleaning may not be effective in preventing the formation of biofilms.
Inadequate Sampling Plans
Poorly designed sampling programs can ignore contamination risks in vital areas.
Incomplete Documentation
Incomplete documents for calibration or verification may lead to serious audit observations.
Lack of Preventive Maintenance
The risk of ignoring maintenance is increased of:
- Equipment failure
- Membrane fouling
- Contamination events
FDA inspections often concentrate on maintenance history and the consistency of operations.
Best Practices for Maintaining GMP Compliance After Validation
Validation isn’t a one-time occasion. Water systems for pharmaceuticals require continual management to ensure compliance for the long term.
Routine Monitoring and Trending
Facilities should be monitored continuously for:
- Conductivity
- TOC
- Microbial levels
- System temperatures
Trend analysis can help identify the gradual degrading of the system.
Preventive Maintenance Programs
Regular maintenance could comprise:
- Membrane replacement
- Sensor calibration
- UV lamp inspection
- Sanitization cycles
- Periodic Requalification
A number of facilities regularly conduct renewal or revalidation to ensure the system’s performance.

Staff Training and SOP Compliance
Operators must be aware of:
- Procedures for sampling
- Sanitization protocols
- Alarm responses
Documentation requirements for GMP
A well-trained staff is essential to keeping the quality of pharmaceutical water in good condition.
