In the world of precision-driven contemporary diagnostics, every milliliter matters. Although clinical laboratories invest billions in high-throughput analyzers, high-purity reagents important “reagent”–water–is often the least understood.
If you’re performing specific immunoassay tests or routine chemistry tests, the quality of pure water for clinical laboratory use serves as the basis for the integrity of your diagnostics. But even the most sophisticated water system for clinical labs can have issues that could affect the accuracy of your results.
Below, we will look at the most frequently encountered five problems with water quality in clinical settings. We also offer solutions that are professional and effective.

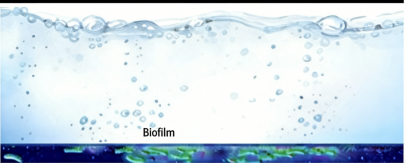
1. Microbial Contamination and Biofilm Formation
Microorganisms represent the main threat to medical testing accuracy. Bacteria exist in water systems because they stick to the internal surfaces of the water system for clinical labs, which form a protective layer called “biofilm.
The Problem: Bacteria produce metabolic by-products that include alkaline phosphatase, which causes direct interference with enzyme-based assays through false-positive and false-negative test results. Bacterial clusters that form physical structures create blockages in the micro-pipetting probes used by automated analyzers.
The Solution:
- 1. UV Sterilization: Your system needs to operate with dual-wavelength UV lamps.
- 2. Recirculation: Bacteria thrive in water that remains still so your system must maintain continuous water flow through its purification system.
- 3. Regular Sanitization: The distribution loop requires chemical sanitization every three months to eliminate emerging biofilms.
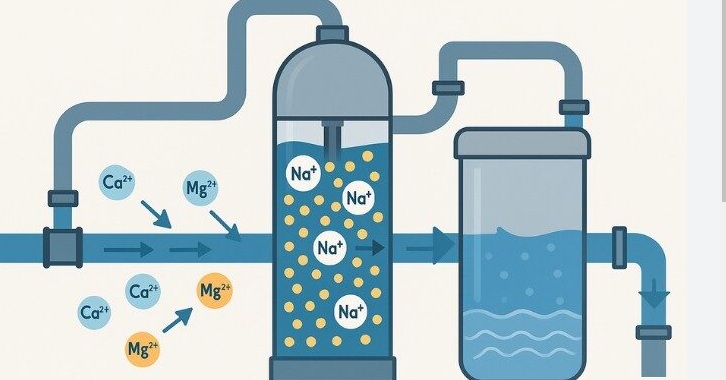
2. Fluctuating Resistivity (Ionic Impurities)
Ionic purity levels in substances can be measured through resistivity which serves as the standard testing method. Clinical procedures need either Type I or Type II water which requires a resistivity of > 10 MΩ·cm.
The Problem: Calcium (Ca²⁺) and Magnesium (Mg²⁺) and Iron (Fe²⁺) ions enter testing areas because they skip the purification process and this leads to disruptions in electrolyte testing and changes in pH meter stability. Your deionization system experiences a complete resin depletion when there is a sudden decrease in resistivity.
The Solution:
- 1.Pre-treatment Optimization: Water softeners should remove “hardness” ions through their high-quality system before the ions reach sensitive membranes.
- 2.Continuous Monitoring: Inline temperature-compensated resistivity meters need to be installed in modern water system for clinical labs which will activate alarms when water quality falls below 10 MΩ·cm.
3. High Total Organic Carbon (TOC) Levels
The Total Organic Carbon (TOC) content of water sources contains organic compounds which include plastic leachables and humic acids.
The Problem: Organics can “ghost” your results by competing with substrates in fluorescence-based assays or by coating the surface of reaction cuvettes, leading to inconsistent light transmission readings. The laboratory needs TOC levels to remain under 50 ppb value because it requires high-sensitivity testing.
The Solution:
- 1. 185nm UV Oxidation: Unlike 254nm lamps, 185nm UV light breaks down organic molecules into CO2 and ions which the system removes through resin.
- 2. Activated Carbon: The pre-filtration stage of your pure water for clinical laboratory setup requires high-density activated carbon which will effectively remove major organic contaminants from the feed water.

4. Particulate Matter and Colloidal Silica
The presence of physical particles in water which appears chemically pure will damage costly laboratory equipment.
The Problem: Silt and sand together with colloidal silica which includes suspended fine particles create mechanical wear on the ceramic pumps and valves inside clinical analyzers. The equipment requires more downtime because of unplanned breaks which generate expensive “preventative maintenance” costs that could have been avoided.
The Solution:
1.Depth Filtration: The system uses 5-micron or 1-micron pre-filters to capture large debris particles.
2.Point-of-Use (POU) Filters: A 0.22μm sterile-grade filter should be installed at the final dispense point of your water system for clinical labs. The analyzer uses this system as its final “safety net” protection.
5.Dissolved Gases (O₂ and CO₂)
The presence of dissolved gases creates an invisible threat which disrupts operations within medical settings.
The Problem: The presence of dissolved oxygen (O₂) causes micro-bubbles to develop in the analyzer’s flow cells which the optical sensor detects as particles. The dissolved carbon dioxide (CO₂) in water creates carbonic acid which decreases pH levels while it also shortens the lifespan of deionization resins.
The Solution:
- Degassing Membranes: High-end systems now incorporate hydrophobic membranes that selectivity remove gases before the water reaches the analyzer.
- Lime Vent Filters: Each clinical laboratory requires its pure water reservoir to have a CO2-absorbent vent filter which stops atmospheric gas from entering the water system.
Ensure Stable Pure Water for Clinical Labs
The process of maintaining consistent water quality requires continuous monitoring and proper system design and routine maintenance instead of being completed through a single installation process. The water system for clinical laboratories will experience performance decline through time because its purification components require regular monitoring and scheduled replacement.
The following best practices require implementation by laboratories to establish dependable pure water systems which clinical laboratory operations need to function properly.
1. Implement Real-Time Water Quality Monitoring
The laboratory water systems of today must deliver uninterrupted tracking for essential parameters which include water resistivity and water conductivity and Total Organic Carbon (TOC) and temperature. These indicators help identify potential contamination early.
A sudden decrease in resistivity indicates that the DI resin has reached its limit while an increase in TOC levels indicates either organic contamination or microbial growth.
2. Follow a Preventive Maintenance Schedule
The purification systems require regular maintenance work to maintain their highest operational efficiency. Laboratories should establish a maintenance plan that includes:
- Timely replacement of RO membranes, DI cartridges, and carbon filters
- Scheduled UV lamp replacement to maintain effective sterilization
- Storage tanks and distribution loops require regular sanitization
- Point-of-use filters need to undergo inspection at established time intervals.
The practice of preventive maintenance safeguards water quality while it increases the operational lifespan of costly laboratory equipment.
3. Design a Closed and Circulating Distribution Loop
A water distribution loop designed properly will stop water from becoming stagnant which helps decrease dangerous microbial contamination. The system maintains water flow because it continuously moves purified water throughout its network.
The atmospheric gases and particles which closed-loop systems exclude from their operations create closed-loop systems that establish conditions for chemical stability and microbial protection.
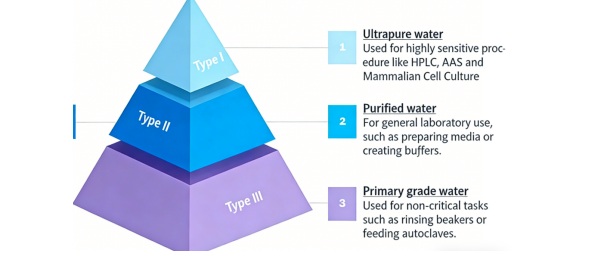
4. Choose the Right Water Grade for Each Application
laboratories use various purified water grades for testing and instrument operation. The CLSI and ASTM international standards create multiple categories for laboratory water which they define as follows:
- Type I water – Ultrapure water used for high-sensitivity assays, molecular diagnostics, and reagent preparation
- Type II water – Suitable for clinical analyzers and general laboratory testing
- Type III water – Typically used as feed water for purification systems
The analytical results of a study depend on selecting the proper water grade which also helps the system function efficiently.
5. Train Laboratory Staff on Water System Management
The laboratory staff members need to learn how to interpret water quality measurements and identify warning alarms from monitoring systems and perform routine sanitization procedures and execute filter and cartridge replacements. The laboratories need proper training for their staff members because this enables them to manage water quality changes which would otherwise impact diagnostic results.
In clinical laboratories, “good enough” water quality presents operational challenges. The five main threats which include microbes and ions and organics and particles and gases can be controlled through professional-grade solutions.
Molewater offers a full range of compliant lab water purification systems from Type III RO to Type I ultrapure water, delivering tailored and reliable solutions for all clinical laboratory water quality needs.




